Alzheimer's disease is the leading cause of neurological disability. It manifests as a progressive and irreversible loss of cognitive functions, particularly memory. Histopathologically, Alzheimer's disease is characterized by the hyperphosphorylation1 of the tau protein and its aggregation in different types of cells. Neurofibrillary degeneration is largely defined by the presence of very dense intracellular tau aggregates called neurofibrillary tangles (NFTs). These latter have been long thought to be a cause of the neurodegeneration seen in patients: indeed their number is strongly correlated with neuronal death and cognitive decline. However, neuronal death and NFTs do not occur in the same zones of the patient's brain. Furthermore, studies performed in transgenic mice have shown that neurons developing NFTs survive and even communicate with one another. Thus, researchers have hypothesized that complex, soluble forms (or "species") of tau with an intermediate level of aggregation (oligomeric forms) may be more toxic than NFTs.
Teams from MIRCen, in collaboration with researchers from Servier, the Lille 2 University in France and the Ludwig-Maximilians University in Germany, set their sights on clarifying the toxicity of tau species in vivo. "We created a 'pro-aggregation' tauopathy model in rats that generates a large number of NFTs in the hippocampus, an area of the brain with a central role in memory," explains Karine Cambon, researcher at MIRCen. Specifically, the team used gene vector technologies to deliver a construct co-expressing wild-type human tau and a pro-aggregation peptide. "With this model, we did observe pathological hyperphosphorylation of tau, mislocalization of the protein in the neurons and the presence of numerous NFTs, similar to what happens in patients, a month after vector delivery," continues Ms. Cambon. "However, by counting the neurons in the hippocampus, we showed that the presence of NFTs did not appear to cause neuronal death, at least up to three months after injection." Surprisingly, in rats receiving wild-type tau without the pro-aggregation peptide, the team observed more greatly hyperphosphorylated tau, a lack of NFTs, and important hippocampal neuronal death.
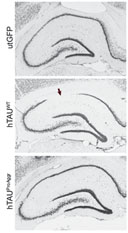
| The toxicity of tau appears to be inversely associated with its ability to aggregate. A specific treatment applied to histological sections of the brain enables the visualization (in black) and count of neurons. After a month of co-expression of wild-type tau and a pro-aggregation peptide (bottom panel), the number of hippocampal neurons remains comparable to the normal hippocampus (top panel). In contrast, after a month of overexpression of wild-type tau alone (middle panel), significant neuronal death occurs in the hippocampus (zone indicated by the arrow). © MIRCen.
|
The results of the study suggest that in vivo, NFTs may cause neurons no harm, at least in the short term, whereas soluble tau species, probably oligomers, appear to be toxic. They furthermore imply that the level of tau hyperphosphorylation may be a more pertinent method of assessing neurodegeneration than NFT count, which only imperfectly reflects disease severity. "Our study suggests that it is surely more appropriate to develop imaging tracers and therapeutic agents targeting specifically these soluble tau species instead of NFTs," concludes Ms. Cambon.
1Phosphorylation is the chemical addition of a phosphoric acid group to a molecule. The phosphorylation of proteins is frequent, usually playing a role in the regulation of their activity.